What is the potential role for pirtobrutinib in the treatment of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL)?
- Pirtobrutinib is a small molecule, noncovalent inhibitor of Bruton’s tyrosine kinase (BTK). BTK is a key signaling protein involved in the activation of pathways for B-cell development, proliferation, and differentiation.1
- There are three other BTK inhibitors (BTKi) approved for CLL/SLL, which include ibrutinib, acalabrutinib, and zanubrutinib. These agents are covalent BTK inhibitors (cBTKi) which bind irreversibly to the C481 residue of BTK. This irreversible binding leads to sustained BTK inhibition, which makes cBTKis highly susceptible to resistance mutations at C481. In contrast, pirtobrutinib is the only approved noncovalent BTK inhibitor (ncBTKi), meaning it binds reversibly to BTK, and it binds to a site that is distant from C481. Therefore, pirtobrutinib retains activity in the presence of C481 mutations.2
- Pirtobrutinib has been incorporated into the second line setting and beyond treatment algorithm based on data published from phase I-II BRUIN trial described below.3
- Pirtobrutinib is a preferred option for patients with intolerance to prior cBTKi or for those with disease that is resistant to cBTKi.
- Pirtobrutinib is also a preferred option for relapsed/refractory (R/R) CLL/SLL after prior therapy with a BTK-based regimen and venetoclax-containing regimen.
- In 2023, the Food and Drug Administration (FDA) granted pirtobrutinib accelerated approval for adults with CLL/SLL who have received at least two prior lines of therapy including a BTKi and a BCL2 inhibitor (BCL2i). This accelerated approval was based on the results from the single arm, phase I-II BRUIN trial.3
- A total of 317 patients with R/R CLL/SLL received pirtobrutinib. The overall response rate (ORR) was 73.3% with a median progression-free survival (PFS) of 19.6 months in patients previously treated with a cBTKi (n=247).
- In patients previously treated with a cBTKi and a venetoclax-containing regimen, the ORR was 70% and the median PFS was 16.8 months (n=100).
- In December 2025, the FDA granted full approval for pirtobrutinib in adult patients with CLL/SLL who have been previously treated with a cBTKi. Compared to the accelerated approval in 2023, the updated FDA-approved indication no longer includes requirements for two or more prior lines of therapy or the need for prior exposure to BCL2i therapy. The full FDA approval is based on results from the phase III BRUIN CLL-321 trial, which is detailed below.3
- The BRUIN CLL-321 trial included 238 patients with CLL/SLL who were previously treated with a cBTKi with or without previous exposure to BCL2i-containing regimens. Patients were randomized 1:1 to receive either pirtobrutinib (n=119) or investigators choice (IC) of idelalisib/rituximab (IdelaR) (n= 82) or bendamustine/rituximab (BR)(n=37).
- All patients had previous exposure to cBTKi (most commonly ibrutinib, 87% of all patients), and 51% of patients had previous exposure to a BCL2i. The median number of therapy lines was three for both treatment arms.
- At a median follow-up of 17.2 months, the primary endpoint of median PFS was 14 months in pirtobrutinib group versus 8.7 months with IC, (HR, 0.54 [95% CI, 0.39 to 0.75]; P =.0002). The PFS benefit seen with pirtobrutinib was observed across clinically relevant subgroups such as patients previously treated with BCL2i, IGHV-unmutated, del(17p)/TP53 mutations, and complex karyotype.
- The 18-month overall survival (OS) rate was 73% in the pirtobrutinib group compared to 71% in the IC arm; however, the OS result is likely confounded due to the high rate of crossover into the pirtobrutinib group, as 76% of IC arm patients crossed over.
- The most common grade 3/4 adverse events in pirtobrutinib group compared to IC were infection (21.6% vs 19.3%), neutropenia (20.7% vs 27.5%), and anemia (11.2% vs 7.3%). Other adverse events of interest in the pirtobrutinib group include atrial fibrillation/flutter, bleeding, and hypertension, which occurred in 2.6%, 21.6%, and 6.9% of patients, respectively.
- Key randomized trials evaluating other guideline-recommended therapies in the relapsed/refractory setting:
- In the phase 3 MURANO trial, venetoclax/rituximab (VenR) was compared with BR in R/R CLL. VenR demonstrated superior efficacy, achieving a median PFS of 54.7 vs 17.0 months and 7-year OS of 69.6% vs 51.0%, with a 7-year PFS of 23%. Only five patients in the trial had previous exposure to BTKi.4
- In phase 3 ASCEND trial, acalabrutinib was compared to IC of IdelaR or BR in patients with R/R CLL. Acalabrutinib showed superior efficacy; at 42 months with PFS rates of 62% in acalabrutinib versus 19% in IC. OS rates were 78% versus 65%, respectively. Patients with previous exposure to BTKi were excluded.5
- In phase 3 ALPINE trial, zanubrutinib was compared to ibrutinib in patients with R/R CLL. At 29.6 months zanubrutinib showed superior efficacy with respect to PFS (HR, 0.65 [95% CI, 0.49 to 0.86]; P=0.002). Patients with previous exposure to BTKi were excluded.6
What role can the pharmacist play in the management of patients on pirtobrutinib?
- Pharmacists play a key role in treatment selection, along with appropriate dosing and dose adjustments, management of adverse effects, and facilitation of medication access.
- Dosing and Administration1
- Recommended dose: 200 mg orally once daily until disease progression or unacceptable toxicity
- Dosage forms and strengths: 50 mg and 100 mg tablets
- Tablets should be stored at room temperature 20°C to 25°C (68°F to 77°F)
- Dose Adjustments for Renal or Hepatic Impairment
- Renal Impairment:
- eGFR ≥ 30 mL/min: No dose reduction needed
- eGFR 15 – 29 mL/min:
- If on 200 mg daily: Reduce to 100 mg once daily
- If on 100 mg daily: Reduce to 50 mg once daily
- If on 50 mg daily: Discontinue therapy
- eGFR < 15 mL/minute: Avoid use
- Hepatic Impairment: No dose adjustments recommended for mild, moderate, or severe hepatic impairment
- Renal Impairment:
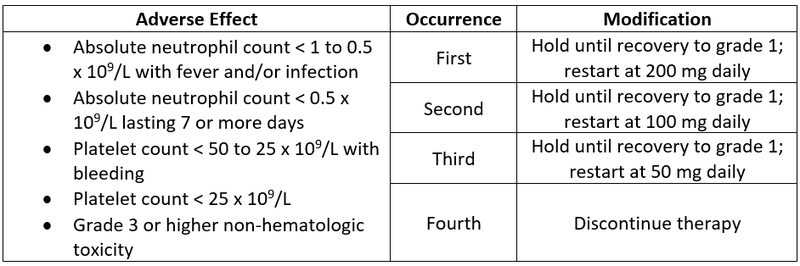
- Dose Modifications for Adverse Effects
- Drug Interactions1
- Concomitant use with strong CYP3A inhibitors
- Avoid concomitant use
- If unable to avoid concomitant use: Reduce dose by 50 mg (i.e. reduce 200 mg to 150 mg)
- If the current dosage is 50 mg once daily, hold pirtobrutinib treatment for the duration of strong CYP3A inhibitor use
- After discontinuation of a strong CYP3A inhibitor for five half-lives, resume pirtobrutinib at the same dose that was taken prior
- Concomitant use with strong or moderate CYP3A inducers
- Avoid concomitant use
- If unable to avoid concomitant use and dose is 200 mg daily, increase to 300 mg daily
- If unable to avoid concomitant use and dose is 50 or 100 mg daily, increase dose by 50 mg
- Pirtobrutinib is a moderate inhibitor of CYP2C8 and BCRP, weak inhibitor of CYP2C19 and CYP3A4, and an inhibitor of P-gp
- Concomitant use with strong CYP3A inhibitors
- Adverse Events1
- Most common adverse reactions of all grades included fatigue (36%), edema (21%), pyrexia (20%), bruising (36%), cough (33%), dyspnea (22%), musculoskeletal pain (32%), COVID 19 (28%), pneumonia (27%), diarrhea (26%), abdominal pain (25%), nausea (25%), hemorrhage (22%), headache (20%).
- In the BRUIN-321 trial, adverse effects led to dose reductions in 3.6%, treatment interruption in 42%, and permanent discontinuation in 9% of patients. Adverse effects that resulted in treatment interruptions in > 5% of patients included pneumonia, neutropenia, febrile neutropenia, and COVID-19.
- Key patient counseling points:
- Advise patients to take pirtobrutinib at the same time each day and to swallow tablets with water. It may be taken with or without food.
- Patients should not cut, crush, or chew tablets.
- If a dose is missed by more than 12 hours, advise patients to skip the dose and take the next dose as scheduled.
- Advise patients to report any signs or symptoms of infection as pirtobrutinib can cause serious infections that may be fatal.
- Inform patients to report any signs or symptoms of bleeding.
- Instruct patients to notify their provider of any upcoming surgeries as pirtobrutinib should be interrupted 3 to 7 days pre and post major surgeries due to risk of bleeding.
- Patient Access – Eli Lilly provides several support programs for pirtobrutinib.10
- JAYPIRCA Saving Card: Co-pay assistance card for commercially insured patients
- JAYPIRCA Interim Access Program: No cost temporary supply for insured, eligible patients who have been prescribed pirtobrutinib for the first time and are experiencing a delay in their insurance coverage

Clinical Pearls
- The following section discusses clinical scenarios that may be encountered in clinical practice:
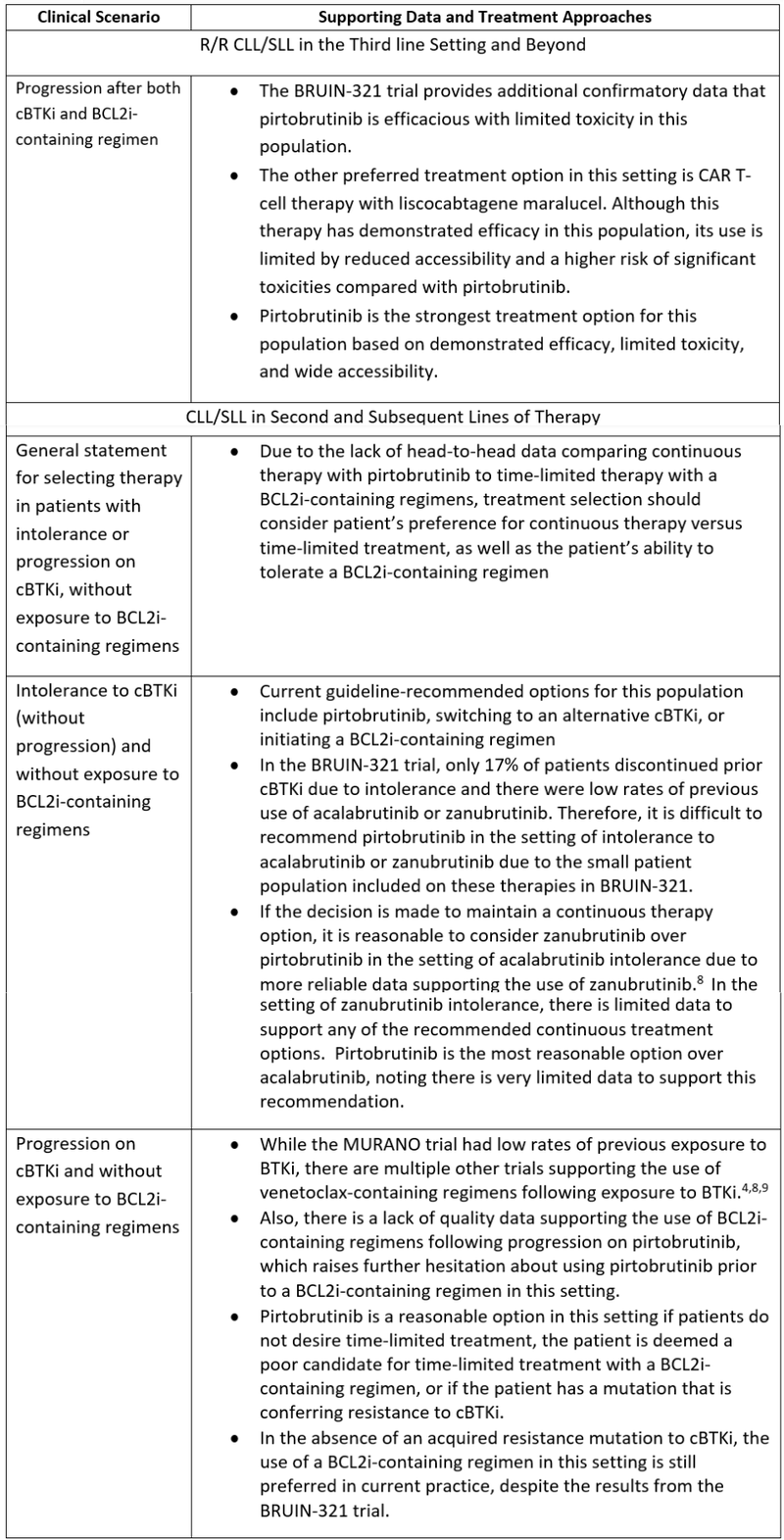
- With the updated FDA-approved indication for pirtobrutinib no longer requiring two prior lines of therapy, optimal sequencing of current therapies for CLL/SLL remains an important area for further investigation. For future trials in this space, it is essential to employ relevant, contemporary comparator arms that reflect evolving standards of care.
- Serious and fatal infections, including bacterial, viral, fungal, and opportunistic pathogens, have been reported in patients receiving pirtobrutinib. Clinicians should consider appropriate vaccination and antimicrobial prophylaxis in patients at increased infectious risk and closely monitor for symptoms, with dose interruption or discontinuation.
References
1.Jayprica (Pirtobrutinib). Package Insert. Eli Lilly and Company; 2025
2.Thompson PA, Tam CS. Pirtobrutinib: a new hope for patients with BTK inhibitor-refractory lymphoproliferative disorders. Blood. 2023;141(26):3137-3142. doi:10.1182/blood.2023020240
3.Sharman JP, Munir T, Grosicki S, et al. Phase III Trial of Pirtobrutinib Versus Idelalisib/Rituximab or Bendamustine/Rituximab in Covalent Bruton Tyrosine Kinase Inhibitor-Pretreated Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (BRUIN CLL-321). J Clin Oncol. 2025;43(22):2538-2549. doi:10.1200/JCO-25-00166
4.Kater AP, Harrup R, Kipps TJ, et al. The MURANO study: final analysis and retreatment/crossover substudy results of VenR for patients with relapsed/refractory CLL. Blood. 2025;145(23):2733-2745. doi:10.1182/blood.2024025525
5.Ghia P, Pluta A, Wach M, et al. Acalabrutinib Versus Investigator's Choice in Relapsed/Refractory Chronic Lymphocytic Leukemia: Final ASCEND Trial Results. Hemasphere. 2022;6(12):e801. Published 2022 Nov 14. doi:10.1097/HS9.0000000000000801
6.Brown JR, Eichhorst B, Hillmen P, et al. Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. N Engl J Med. 2023;388(4):319-332. doi:10.1056/NEJMoa2211582
7.Shadman M, Burke JM, Cultrera J, et al. Zanubrutinib is well tolerated and effective in patients with CLL/SLL intolerant of ibrutinib/acalabrutinib: updated results. Blood Adv. 2025;9(16):4100-4110. doi:10.1182/bloodadvances.2024015493
8.Jones JA, Mato AR, Wierda WG, et al. Venetoclax for chronic lymphocytic leukaemia progressing after ibrutinib: an interim analysis of a multicentre, open-label, phase 2 trial. Lancet Oncol. 2018;19(1):65-75. doi:10.1016/S1470-2045(17)30909-9
9.Eyre TA, Kirkwood AA, Gohill S, et al. Efficacy of venetoclax monotherapy in patients with relapsed chronic lymphocytic leukaemia in the post-BCR inhibitor setting: a UK wide analysis. Br J Haematol. 2019;185(4):656-669. doi:10.1111/bjh.15802
10.Lilly. Savings & Support – Jaypirca. Eli Lilly and Company. https://jaypirca.lilly.com/hcp/savings-support#savings-card-download