Authors
- Sanja Zepcan, PharmD, BCPS, BCOP, Clinical Pharmacy Specialist Hematology/Oncology Bone Marrow Transplant and Cellular Therapy, Loyola University Medical Center, Chicago, IL
- Emily Rux, PharmD, BCOP, Hematology/Oncology Clinical Pharmacist & Clinical Lead Pharmacist, Loyola University Medical Center, Chicago, IL
Introduction & Why MajesTEC-3 Matters
Since quadruplet therapy redefined first line therapy for multiple myeloma (MM), investigation into the optimal second line therapy has been an active field of research opening many options for patients after initial relapse.1 Second-line treatment is no longer a passive extension of frontline strategies, but a dynamic space marked by earlier integration of cellular therapies and re-emergence of an old drug, belantamab-mafodotin.2–4 This space now has breakthrough data with BCMA-directed CD3 T-cell engager teclistamab-cqyv plus daratumumab hyaluronidase-fihj (Tec-Dara), presented at ASH in December of 2025 and shortly after that was published in the New England Journal of Medicine.2
MajesTEC-3 is the first phase III trial to demonstrate both progression-free (PFS) and overall survival (OS) benefits with a BCMA×CD3 bispecific antibody plus anti-CD38 therapy in earlier relapsed disease.2 While these results are practice-changing, they also raise clinical questions regarding optimal treatment sequencing, operational feasibility, safety considerations, and applicability to the current therapy landscape with quadruplet therapy used as a standard of care in the first line setting. Nevertheless, this paradigm shift toward earlier use of bispecific antibodies, further underscored by the National Priority Review Voucher awarded to the Tec-Dara combination, which accelerated regulatory review and reshaped the relapsed MM treatment algorithm.5 Most recently, the FDA approved teclistamab in combination with daratumumab hyaluronidase-fihj on March 5, 2026, for adults with relapsed or refractory multiple myeloma who have received at least one prior therapy that included a proteasome inhibitor and an immunomodulatory agent.6
Background
Teclistamab is a bispecific T-cell engager that binds to B-cell maturation antigen (BCMA) expressed on multiple myeloma cells and CD3 expressed on the surface of T cells to engage killing of MM cells.7 Teclistamab is currently approved in relapsed/refractory multiple myeloma (RRMM) after ≥4 prior lines. Daratumumab is a CD38-directed monoclonal antibody, available as an intravenous as well as subcutaneous formulation for administration, with multiple indications for MM, now widely used in the first line setting.8 The plausible rationale for combining teclistamab and daratumumab is based on complementary and potentially synergistic immunologic mechanisms. Dual targeting of BCMA and CD38 may enhance immune activation which is designed to improve outcomes early in the relapsed setting.9 Dual targeting enables simultaneous engagement of malignant plasma cells through distinct surface antigens while enhancing immune-mediated tumor killing.10 Daratumumab depletes CD38-expressing immunosuppressive cells, including regulatory T cells, regulatory B cells, and myeloid-derived suppressor cells, thereby augmenting T-cell fitness and function, inducing durable increases in helper and cytotoxic T-cell absolute counts.10 This immune modulation may enhance the activity of T-cell–redirecting therapies such as teclistamab. Together, this strategy is designed to amplify antitumor immune activation, deepen responses, and improve clinical outcomes when deployed earlier in the relapsed disease setting.10
MajesTEC-3 Overview
MajesTEC-3 was a phase 3, multicenter, open-label trial (NCT05083169) conducted across 150 sites in 20 countries between October 22, 2021, and September 23, 2023. Patients were randomized 1:1 to receive teclistamab plus daratumumab or investigator’s choice of either daratumumab, pomalidomide, and dexamethasone (DPd) or daratumumab, bortezomib, and dexamethasone (DVd).2 Eligible patients had received one to three prior lines of therapy, including a proteasome inhibitor and lenalidomide, with documented progression. Patients with only one prior line of therapy required proof of lenalidomide-refractory myeloma. Key exclusion criteria included prior BCMA-directed therapy or patients shown to be refractory to anti-CD38 monoclonal antibodies. Patients shown to be refractory to a specific agent were not randomized to receive treatment that included that agent.
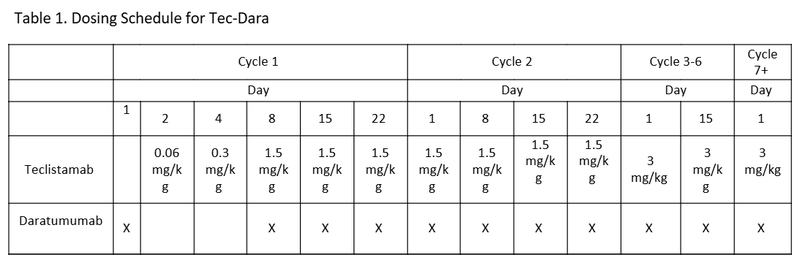
The teclistamab plus daratumumab group (Tec-Dara) cohort received teclistamab as two step up doses in week 1 of cycle 1, followed by 1.5 mg/kg weekly for the reminder of cycle 1 and 2. The dose increased to 3 mg/kg every other week for cycles 3-6, and 3 mg/kg monthly from cycle 7 onward (Table 1). The DPd and DVd groups received treatment according to established local institutional protocols. Treatment in each group was continued until progressive disease, death, unacceptable adverse events, or withdrawal of consent.
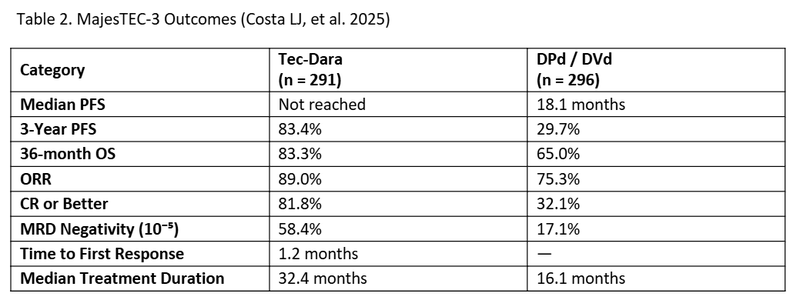
A total of 587 patients were randomized (Tec-Dara n=291 and DPd/DVd n=296). Baseline characteristics were similar between groups. The median time to first response and time to occurrence of complete response (CR) or better was 1.2 months (0.2-5.5) and 6.9 months respectively. The median duration of treatment with Tec-Dara was longer compared to DPd or DVd (32.4 months vs. 16.1 months). Tec–Dara demonstrated a significant improvement in PFS compared with standard regimens, with 3-year PFS rates of 83.4% versus approximately 29.7% (hazard ratio [HR], 0.17; P<0.0001). After 36 months of follow up, median progression free survival (PFS) was not reached in the Tec-Dara arm versus 18.1 months in the DPd/DVd arm. Overall response rate (ORR) was higher (89%) in Tec-Dara compared to the DPd/DVd arm (75.3%). A significant overall survival (OS) benefit was also observed (HR, 0.46; P<0.0001), with 36-month OS rates of 83.3% with Tec-Dara versus 65.0% with DPd/DVd arm. Deeper responses were achieved with Tec–Dara, including higher rates of CR or better (81.8% vs 32.1%), as well as substantially higher rates of minimal residual disease (MRD) negativity (58.4% vs.17.1%). Overall response rates (ORR) was also significantly higher with Tec-Dara (89.0% vs 75.3%). Response rates were lower in patients with extramedullary disease, stage III disease, and patients with >/= 60% plasma cells in their bone marrow. Alternatively, response rates were higher in patients who had received three or less prior lines of therapy.
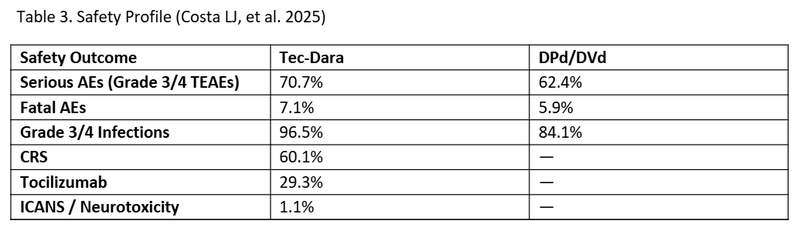
Similar overall rates of grade 3/4 treatment-emergent adverse events (TEAEs) occurred in both arms. Serious adverse events (Grade 3/4) were more frequent in the Tec–Dara arm (70.7%) than in the DPd/DVd arm (62.4%); however, fatal adverse events occurred at comparable rates (7.1% vs 5.9%, respectively). Grade 3/4 infections were more frequent in the Tec-Dara arm (96.5% vs 84.1%) but declined with immunoglobulin supplementation and prophylaxis protocols for Pneumocystis jirovecii pneumonia and herpes zoster reactivation. This confirms the clinical importance of IgG levels monitoring and protocolized IVIG administration. Per final Protocol Amendment 9 of Study: MajesTEC-3 (MMY3001) patients received either prophylactic IVIG prior to the first dose of therapy or initiated replacement therapy prior treatment for IgG levels <400mg/.11 Cytokine release syndrome (CRS) occurred in 60.1% of patients with most occurring after step up and cycle 1 doses. All CRS cases were grade 1 or 2 in severity. The median time to onset of CRS was 2 days (1-22). Tocilizumab was allowed for grade 1 CRS. Neurotoxic events, including immune effector cell-associated neurotoxicity syndrome (ICANS), were rare (n =3); two patients had grade 1 and one patient had grade 4. No patients discontinued therapy due to CRS, while 1 patient discontinued due to ICANS.
Clinical Impact
MajesTEC-3 has the potential to establish a new standard of care in the second-line setting. The combination of Tec–Dara delivers efficacy approaching that of BCMA CAR-T therapy, including high ORR, deep responses, and prolonged PFS, while also demonstrating a proven OS benefit. Importantly, Tec–Dara avoids the unpredictable neurologic and prolonged hematologic toxicities associated with CAR-T therapy and offers the advantage of being an off-the-shelf regimen which can be administered at community centers experienced with bispecific T-cell engagers.12 Despite these strengths, the relevance of MajesTEC-3 in the modern treatment era warrants careful consideration. Only approximately 15% of enrolled patients had prior exposure to daratumumab, and there is currently no data supporting the use of Tec–Dara following frontline quadruplet therapy, which incorporates anti-CD38 antibodies.2 CAR-T therapy remains a one-time treatment that offers patients the possibility of extended treatment-free intervals, whereas in contrast Tec–Dara is administered continuously until disease progression.2,3 MajesTEC-3 also underscores the importance of routine immunoglobulin monitoring and proactive IVIG supplementation to mitigate infection risk in patients receiving bispecific antibodies, highlighting evolving safety considerations for this medication class.2
As multiple BCMA-targeted strategies move into earlier lines of therapy across different trials, treatment selection after first relapse should be individualized, taking into account patient characteristics, comorbidities (including baseline neurologic risk), caregiver support, geographic access to specialized centers, and institutional experience. In this increasingly complex landscape, both bispecific antibodies and CAR-T therapies represent complementary, rather than competing, approaches to optimizing outcomes in relapsed multiple myeloma. An important consideration when sequencing BCMA-targeted therapies is the potential impact of prior BCMA exposure on subsequent treatment efficacy. In the phase 2 CARTITUDE-2 trial, the efficacy of CAR T-cell therapy appeared reduced in patients with prior BCMA-directed therapy, with response rates decreasing from approximately 85–95% to about 60%.13

Conclusion
Current treatment guidelines have been updated to reflect this data, recommending the use of teclistamab plus daratumumab after first relapse in patients with disease refractory to both bortezomib and lenalidomide.1 While we are awaiting longer follow-up, the important questions remain regarding the durability of the overall survival benefit and long-term safety with prolonged exposure to bispecific antibody therapy. In parallel, three ongoing studies, MajesTEC-4, MajesTEC-5, and MajesTEC-7, are exploring BCMA-directed bispecific antibodies in combination regimens across earlier disease settings, including maintenance and frontline therapy.
Collectively, MajesTEC-3 represents a practice-changing milestone in multiple myeloma, providing the first phase III evidence that a BCMA×CD3 bispecific antibody combined with anti-CD38 therapy can deliver meaningful improvements in both progression-free and overall survival when used early in relapse. As BCMA-directed therapies continue to move across lines of treatment, clinicians must interpret these results within an increasingly complex myeloma landscape - balancing efficacy, toxicity, logistics, and sequencing - to individualize therapy and optimize long-term outcomes.
References
1.NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Multiple Myeloma. Version 5.2026. https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
2.Costa LJ, Bahlis NJ, Perrot A, et al. Teclistamab plus Daratumumab in Relapsed or Refractory Multiple Myeloma. N Engl J Med. Published online December 9, 2025:NEJMoa2514663. doi:10.1056/NEJMoa2514663
3.San-Miguel J, Dhakal B, Yong K, et al. Cilta-cel or Standard Care in Lenalidomide-Refractory Multiple Myeloma. N Engl J Med. 2023;389(4):335-347. doi:10.1056/NEJMoa2303379
4.Hungria V, Robak P, Hus M, et al. Belantamab Mafodotin, Bortezomib, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2024;391(5):393-407. doi:10.1056/NEJMoa2405090
5.Food and Drug Administration. FDA proactively awards national priority voucher based on strong phase 3 study results. December 15, 2025. Accessed December 15, 2025. https://www.fda.gov/news-events/press-announcements/fda-proactively-awards-national-priority-voucher-based-strong-phase-3-study-results
6.Food and Drug Administration. FDA approves teclistamab in combination with daratumumab hyaluronidase-fihj for relapsed or refractory multiple myeloma. March 5, 2026. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-teclistamab-combination-daratumumab-hyaluronidase-fihj-relapsed-or-refractory-multiple
7.Tecvayli (teclistamab-cqyv) injection, for subcutaneous use. Accessed January 15, 2026. https://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/TECVAYLI-pi.pdf
8.Darzalex (daratumumab) injection, for intravenous use; Darzalex Faspro (daratumumab and hyaluronidase-fihj) injection, for subcutaneous use. Accessed January 15, 2026. https://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/DARZALEX-pi.pdf
9.Van De Donk NWCJ, Richardson PG, Malavasi F. CD38 antibodies in multiple myeloma: back to the future. Blood. 2018;131(1):13-29. doi:10.1182/blood-2017-06-740944
10.Krejcik J, Casneuf T, Nijhof IS, et al. Daratumumab depletes CD38+ immune regulatory cells, promotes T-cell expansion, and skews T-cell repertoire in multiple myeloma. Blood. 2016;128(3):384-394. doi:10.1182/blood-2015-12-687749
11.Costa LJ, Bahlis NJ, Perrot A, et al. Trial protocol for: Teclistamab plus daratumumab in relapsed or refractory multiple myeloma. Supplement to: N Engl J Med. 2026;394:739-752. doi:10.1056/NEJMoa2514663.
12.Lim KJC, Tan M, Parrondo R, et al. Clinical course, risk factors and mitigating strategies for Immune effector cell-associated late onset neurotoxicities after ciltacabtagene autoleucel CAR-T in multiple myeloma. Blood Cancer J. 2025;16(1):18. doi:10.1038/s41408-025-01441-3
13.Cohen AD, Mateos MV, Cohen YC, et al. Efficacy and safety of cilta-cel in patients with progressive multiple myeloma after exposure to other BCMA-targeting agents. Blood. 2023;141(3):219-230. doi:10.1182/blood.2022015526
14.Einsele H, San-Miguel J, Dhakal B, et al. Cilta-cel in lenalidomide-refractory multiple myeloma (CARTITUDE-4): an updated analysis including overall survival from an open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2026;27(2):254-268. doi:10.1016/S1470-2045(25)00653-9
15.Hungria V, Robak P, Hus M, et al. Belantamab mafodotin plus bortezomib and dexamethasone in patients with relapsed or refractory multiple myeloma (DREAMM-7): updated overall survival analysis from a global, randomised, open-label, phase 3 trial. Lancet Oncol. 2025;26(8):1067-1080. doi:10.1016/S1470-2045(25)00330-4